Entering and editing data in a PMS study
Overview
Note! When editing this lesson, make sure to check if the corresponding edit needs to be made in Clinic>Data entry>Entering and editing data.
The subject Details page
When you select a subject card on the Selection page, or add a new subject, the subject Details page opens:
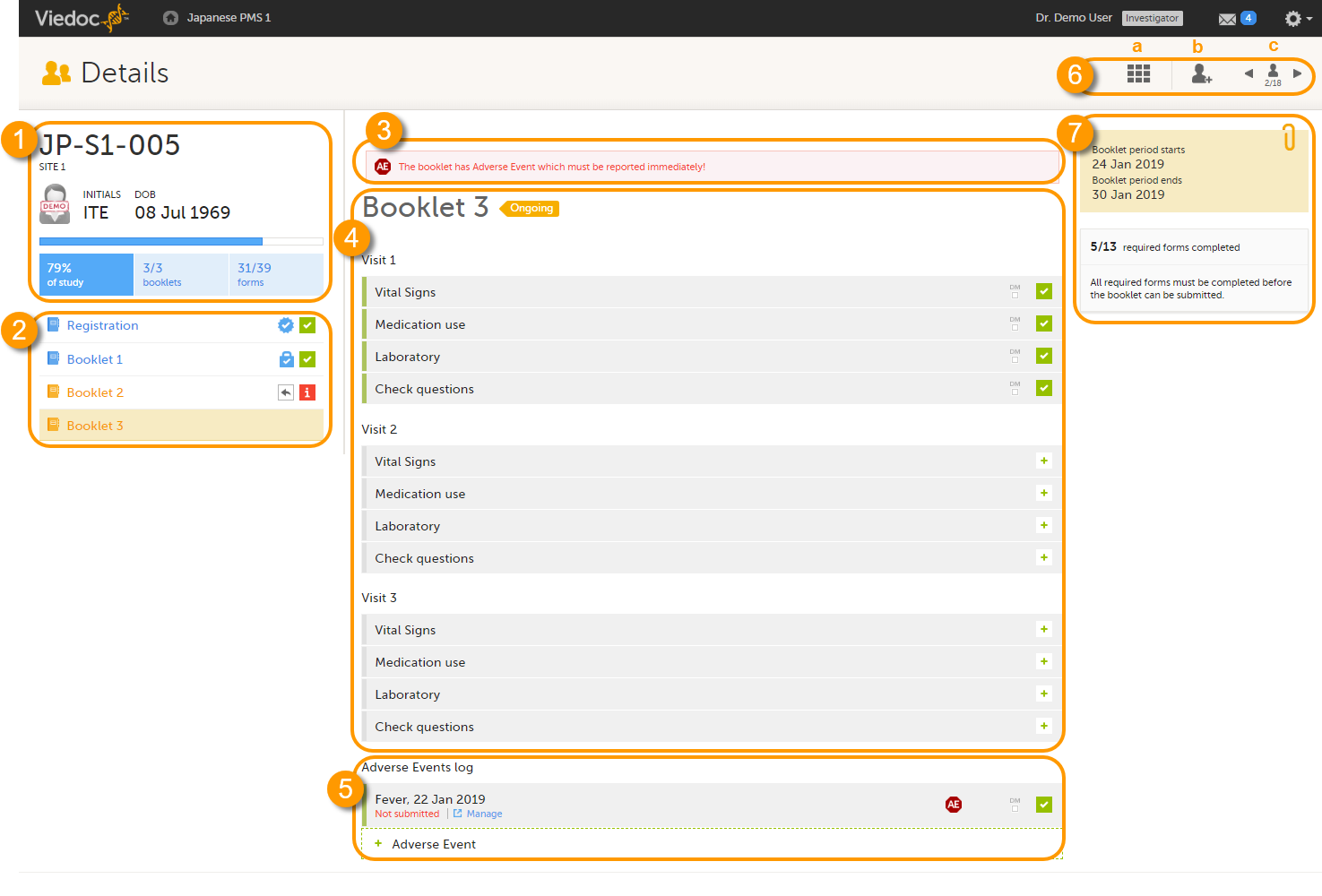
The details page provides the following information:
1. Subject details - including subject ID, site name and a status summary.
2. Booklet overview - a list of all booklets for the subject. Booklets are displayed in grey if the time period for the booklet has not started yet. Booklets are displayed in orange if they are open (the current time point is within the active period of the booklet), or if they are recalled or returned, and hence are open for data edit at the clinic side. Booklets are displayed in blue if they are submitted to the sponsor side and thus closed for data edit at the clinic side. For more information about the submit/recall/receive/return process of handling booklets, see Overview of the submit-receive-return process.
Icons behind the booklet show the status of the forms in the booklet, as follows:
| Icon | Description |
|---|---|
 |
Tasks - open tasks for the sponsor side user (Data Manager (DM) review, approving a query), the number of open tasks is indicated in the icon |
 |
Submitted - the booklet is submitted to the sponsor (the icon does not indicate whether the booklet has been received or not) |
 |
Returned - the booklet has been returned from the sponsor side to the clinic side |
 |
Frozen - the booklet has been frozen on the sponsor side, and is locked for data edit. |
 |
Issue - at least one open query and/or missing data |
 |
Complete - all events and forms in the booklet have been completely filled in |
3. If the booklet contains an Adverse Event (AE), a warning message will appear here stating that the booklet has AEs that must be reported immediately.
4. The list of events and forms to be filled in for the selected booklet. Click on the form bar to open it and enter the data. See Enter data.
Icons on the form show the status of the forms in the booklet, as follows:
| Icon | Description |
|---|---|
 |
Tasks - open tasks for the sponsor side user (DM review, approving a query), the number of open tasks is indicated in the icon |
 |
Signed - the data in the form has been signed by the investigator (forms are automatically signed when the booklet containing them is submitted) |
 |
DM review - the data are not yet reviewed by the sponsor side user |
 |
DM review - the data are reviewed by the sponsor side user |
 |
Issue - at least one open query and/or missing data |
 |
Complete - all events and forms in the booklet have been completely filled in (all required data items are completed) |
 |
Adverse Event - this form contains an adverse event |
5. The AE log, in case AEs have been added.
6. Toolbar with the following functions:
a. return to the Selection page.
b. add a new subject.
c. toggle between subjects.
7. The time period when the booklets starts and ends, and the number of required forms that are completed, depicted as X/Y, where X is the number of completed forms and Y is the total number of required forms.
See also the video tutorial Enter data.
An example of a form
When you click a form on the subject Details page, the form opens. The below image provides an example of a form.
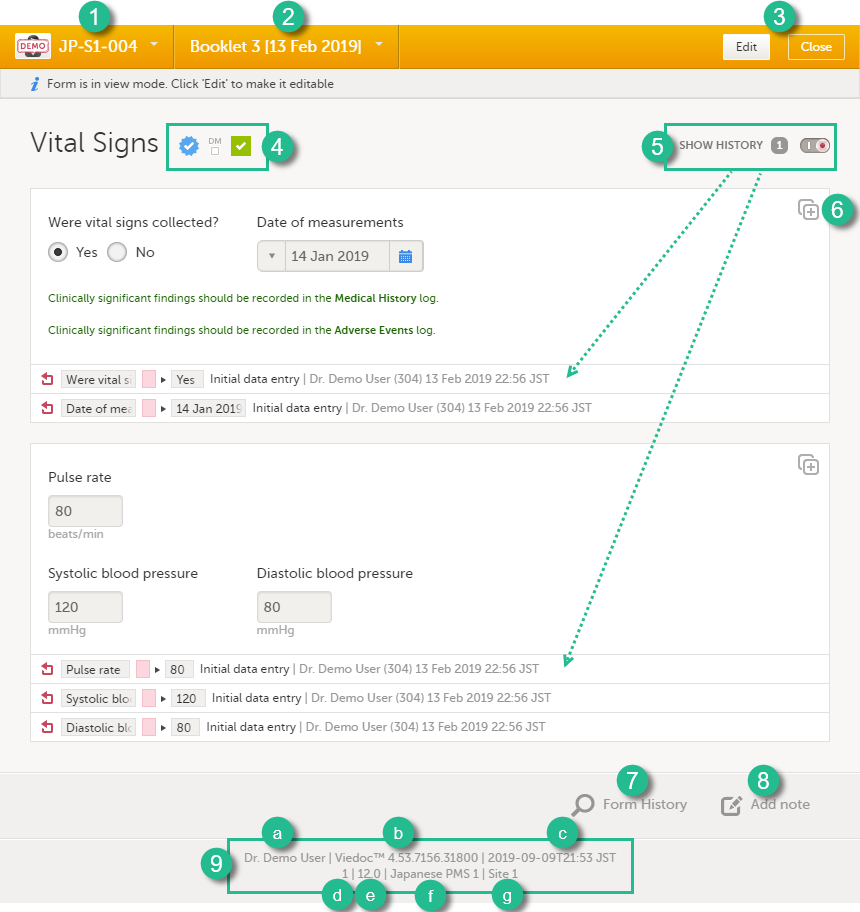
1. Subject ID. You can use this drop-down list to navigate to other subjects within the same form, see below for more information.
2. Booklet name. You can use this drop-down list to navigate to other booklets within the same form, see below for more information.
3. Close. Click to close the form and return to the subject Details page.
4. Icons showing the status of the form, see the description of these icons above.
5. Show history. Activate the Show history switch to display the history of each form item.
6. Action button. Click to select a field (item) and select the type of action you wish to perform, for example to confirm data as missing or to add a private note. For more information Confirm data as missing and Private notes.
7. Form History, see Form history PDF below for more information.
8. Add note. Click here to add a private note to the form, see Private notes for more information.
9. Footer of the form, contains the following information:
a. the user name of the user
b. Viedoc version number
c. date and time of last form edit
d. the protocol version that the study is based on
e. study design version number of the version that is active on the moment the event is initiated
f. name of the study
g. name of the site
Initiating a booklet
Note! A booklet can only be initiated when the start date of the booklet has been reached, that is, if the booklet is displayed in orange text. If the booklet is displayed in grey text, it is not possible to initiate the booklet.
To initiate a booklet:
| 1 |
In the booklet overview pane on the left side of the subject Details page, select the booklet you would like to initiate. |
| 2 |
Click any form and fill in the required data. |
| 3 | Click Save changes. The form closes and the booklet is initiated. The booklet status will change from Not initiated to Ongoing. |
Entering data
Working in multiple browser tabs
Important! Working in multiple browser tabs when entering data may cause data conflicts and other serious problems. Therefore it is important to only work in one browser tab when entering or editing data.
Entering data in a form
Open a form by clicking the form bar, and complete the questions in the form.
Most fields are self-explanatory when it comes to how they should be filled in. Below, we explain how to fill in fields for:
When all the forms in one event are filled in, the event is considered completed and a green check mark appears on the event tab in the event slider.
Dates
You can fill in the date field in two different ways:
- Click the drop-down menu on the left side of the date field, and select one of the following options:

- event date current date (and time)
- yesterday
- time not known
- day not known
- month not known
- Click the calendar icon to open the date picker and select a date, or:
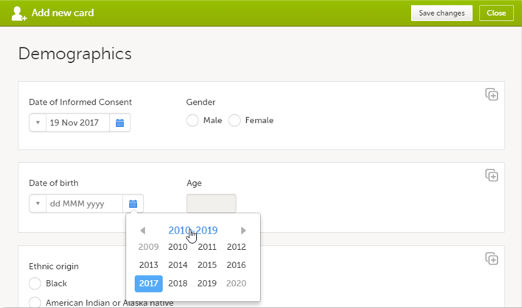
- click the month header to view all the months of the year
- click the year header to view a range of years
- then select the year, month and day
To remove a date, click the drop-down menu on the left side of the date field and select clear.
Times
You can fill in the time field in two different ways:
- Click the drop-down menu on the left side of the date field to select the current time:
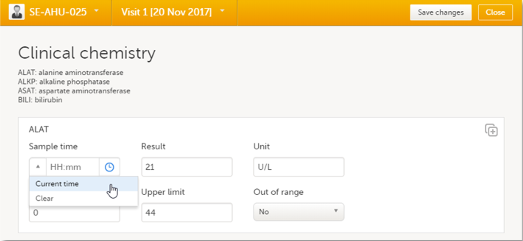
- Click the clock icon on the right side of the time field to open the time picker. Click the arrows to select the hour and minute (with a five minutes time interval), or double-click the hour and minute fields to manually enter any hour and minute:

Range
You can define a range of values by selecting a comparator symbol, and then entering the numeric value(s):
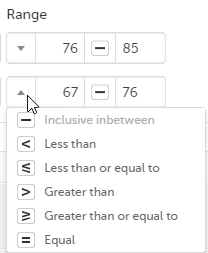
The following comparators are available:
- “-“ – Inclusive in between
- “<” – Less than
- “≤” – Less than or equal to
- “>” – Greater than
- “≥” – Greater than or equal to
- “=” – Equal
File upload
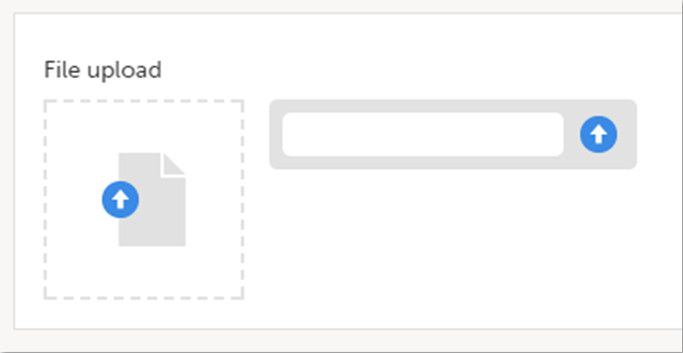
To upload a file to a form:
| 1 | Click the arrow on the right side of the upload box or click the thumbnail if this is available. |
| 2 | Browse for the file to be uploaded and click Open. During the upload process:
|
| 3 | The uploaded file(s) will be stored once the respective form is saved by clicking Save changes. |
You can download an existing file by clicking the file name or by clicking the thumbnail (if it exists).
You can remove an uploaded file by clicking the trash can icon on the right side of the file name.
Notes!
- The file upload icon will display a thumbnail of the image if a jpeg, gif, or png file is uploaded. If other file types are uploaded, the icon will only show the file extension.
- The maximum allowed file size is 2 GB.
- The upload of password-protected zip files is not supported, as Viedoc cannot scan these files for viruses.
- For security reasons, it is not possible to upload executable files. See the complete list of unsupported file types in the end of this lesson, in Blacklisted file formats.
Linking between forms
Depending on your study configuration, when editing a form, you can add links between different types of forms with the Form link item. You can also submit a form that links to another form. This can be useful for example, if you want to add complementary data to the submitted form. You can link to a form regardless if it is a form that can be submitted individually or not.
When a linking form (for example an AE) is linked to another form- the linked form, (for example the Prior and Concomitant Medications form), and the AE form is submitted, a read-only copy of the Prior and Concomitant Medications form is sent to the sponsor side (if it is not already available on the sponsor side). For more information, see Linked form copy
Notes!
- To access the Form link item, Clinic users must have access to whichever form type it is configured to link to, for example the Adverse Events, Medical History, or Prior and Concomitant Medications forms.
- Form link items cannot be mapped to an item in a repeating item group.
Forms can be linked with several instances of a specified form type. In this example, the Prior and Concomitant Medications form can be linked to one or more Adverse Event forms.
To link the Adverse Event form to a Prior and Concomitant Medications form:
| 1 |
Open a subject card. In this card you can see a Prior and Concomitant Medications form under the Registration booklet: 
In this example, we will link an Adverse Event form to a Prior and Concomitant Medications form. To link an Adverse Event form to a Prior and Concomitant Medications form, select the Adverse Events form from the Details page. 1. The Adverse Event form opens. The available link items are shown in a dropdown list. 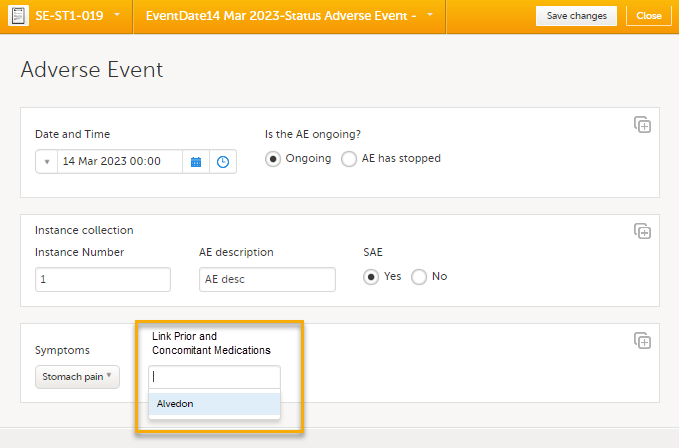
2. Select the link item and Select Save changes - the Adverse Event form is now linked to the Prior and Concomitant Medications form instance. Note! The forms in the dropdown list of link items are shown in the order of the earliest date first according to the event date, (and by the order of activities reflecting the order in the design) within each event. |
| 2 |
After saving the form, select any link item to open and read that form. In the example shown below, the Prior and Concomitant Medications form was the link item. Closing the form returns you to the original form, in this example, the Adverse Event form. 
Note! You can also search in the form link item field. This can be useful to find a specific form instance if there are many adverse events for example. |
Updates to linked forms
If you update a linked form by resetting, deleting, or changing data, the following occurs:
- The form containing the linked form instance is marked with an issue flag (the red [ i ] icon).
- The following error message is displayed at the top of the form containing the link item:

If you then select Edit on the linking form, that form is automatically updated with the latest updates from the linked form.
Locations of updated linked forms
In the Issues view of the Selection page, forms are easily identified and are labelled Form link broken:
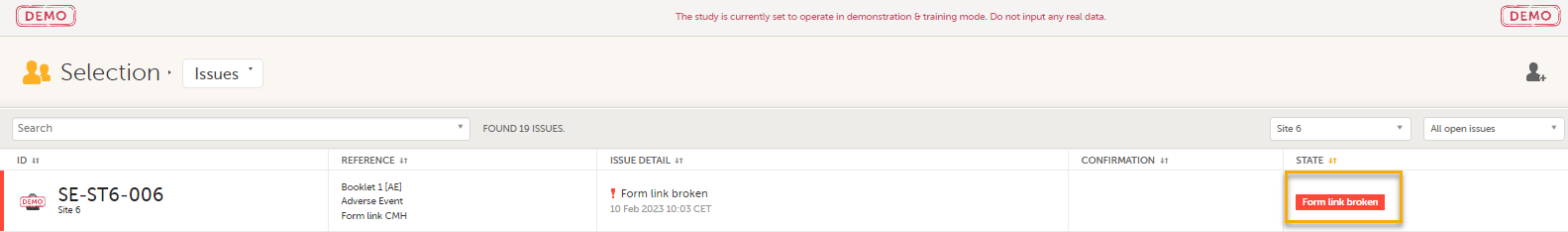
On the subject card, the issue is marked with an issue flag:
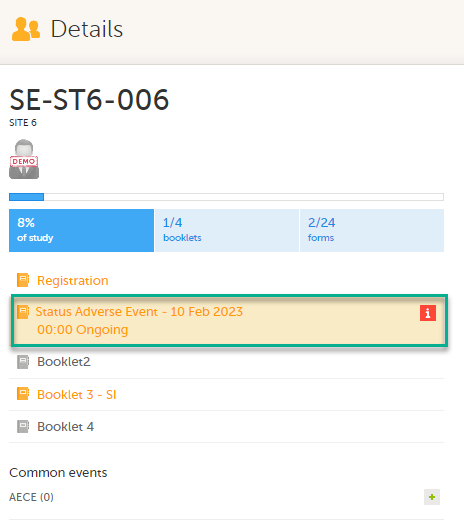
Updating a linked form
To update a linked form:
| 1 |
Select the booklet marked with the red [ i ] icon to open the issue, (in this example, the Adverse Event). 
|
| 2 |
Select the form with the red [ i ] icon to open the form with the issue. |
| 3 |
Select Edit to update the form: 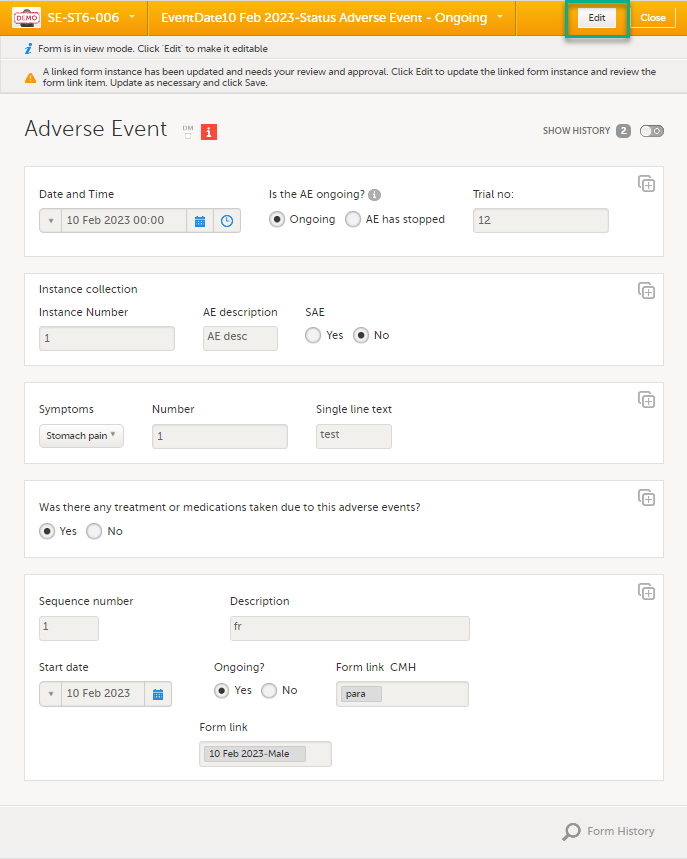

|
| 4 |
A message is displayed as part of the audit trail when the linked items are updated: 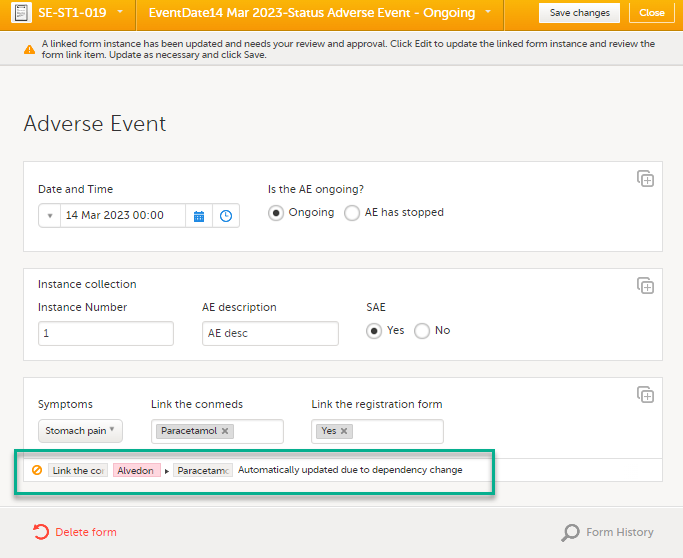
|
Linked form copy
When a form (for example an AE) is linked to another form, (for example the Prior and Concomitant Medications form), and the AE form is submitted, a read-only copy, labelled Form copy submitted, of the Prior and Concomitant Medications form is sent to the sponsor side (if it is not already available). The linked form is locked for editing once it has been submitted as a copy, until it has either been received on the sponsor side,or recalled by the clinic side.
Note! If a booklet contains a form that is linked to another form within that same booklet, the linked form is NOT submitted.
For more information about submitting and recalling linked forms, see Submitting and recalling booklets, AE reports and forms.
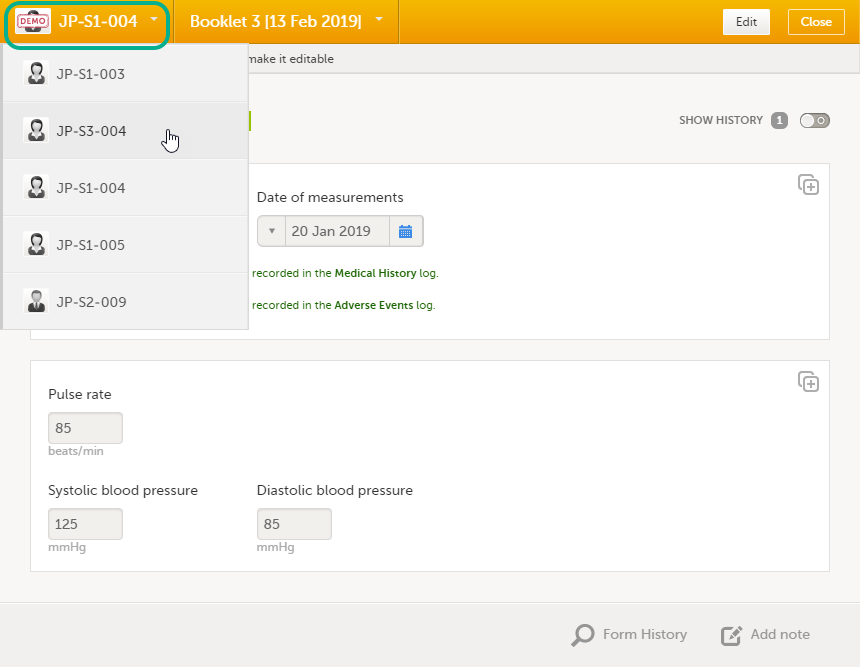
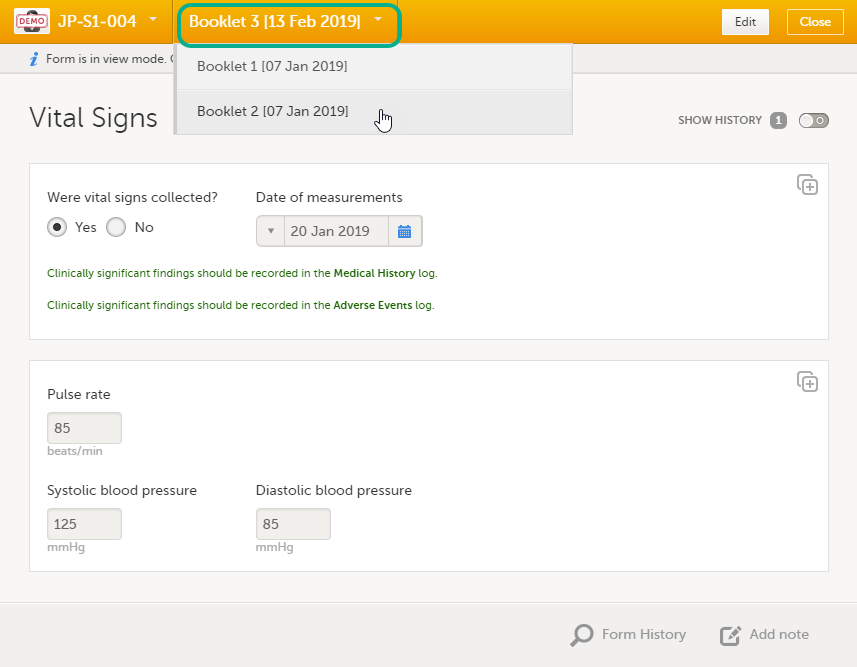
Navigating between subjects/booklets within same form
Once you have a form open, it is possible to navigate through different subjects, or different events, if the respective form is included in other initiated events as well. This is available through the drop-downs in the form header, as illustrated below:
- Navigate through subjects:
- Navigate through booklets:
Editing data
To edit data that already have been saved:
| 1 | Open the form that contains the data you want to change. |
| 2 | Click Edit in the top right corner of the form: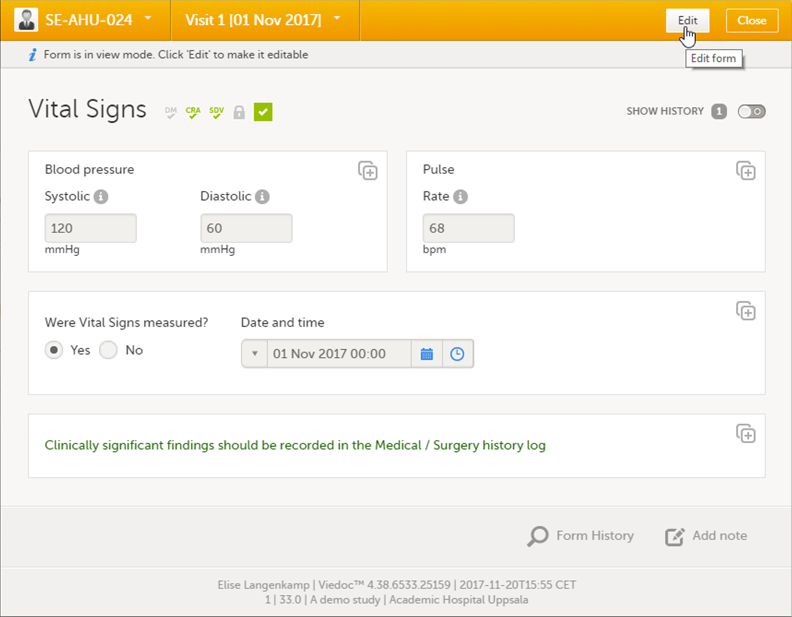 |
| 3 | Edit the data and click Give reason. A pop-up will be displayed: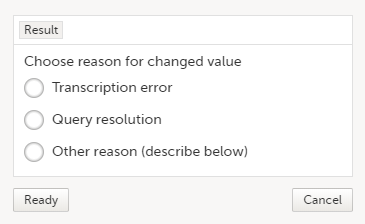 |
| 4 | Select one of the pre-formulated reasons for change, or click Other reason and provide a description. Click Ready, and click Save changes. |
Note!
- A form that has been locked by the monitor or data manager cannot be edited. If you still want to edit a locked form, contact your local Monitor.
- For scheduled and unscheduled events, when the event date form ($EVENT) is excluded when you use automatic event dates, it still counts. In the signing console the counter (number of forms) for a event includes the $EVENT form. It cannot be selected to be signed but can be signed if you select sign all (for subject or event). If you sign forms on an event individually you will not be able to sign the $EVENT form. This in turn makes it so that the sign symbol that appears on the event when everything is signed does not appear, even though it looks like everything is signed.
Repeating forms
It is possible to create several instances of the same form within one activity. This can only be done if the form is set as repeating in the study design.
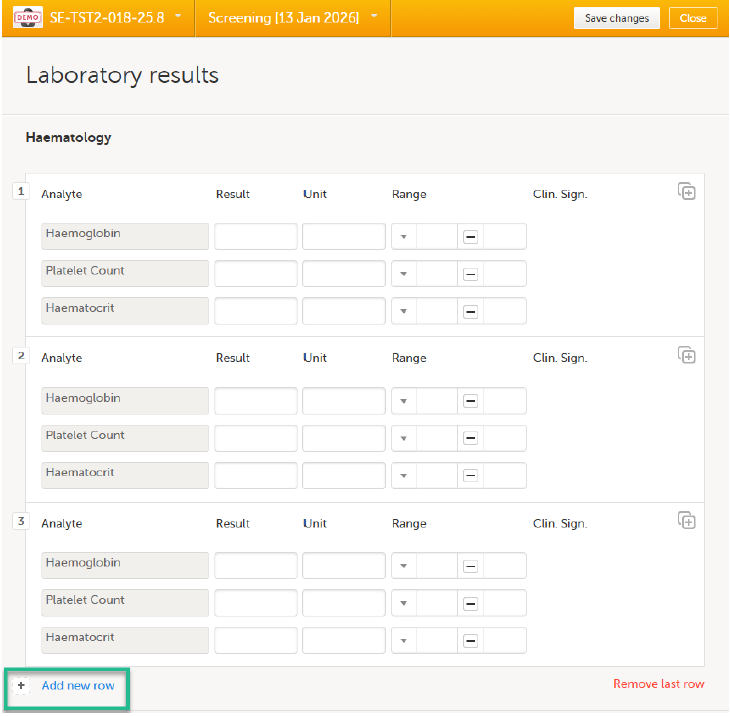
If a form is set as repeating, a ghost form will appear once you have filled out and saved the first instance of the form (see the lab form example in the image).
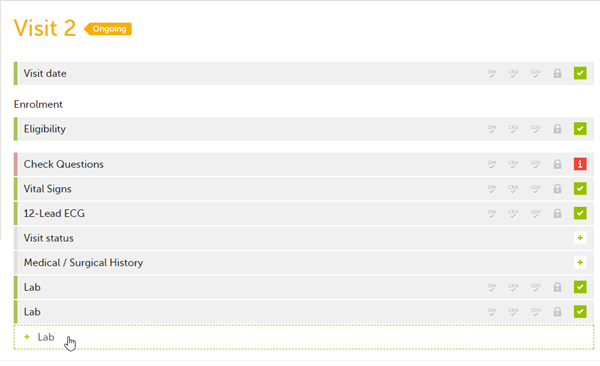
To fill in an instance of a repeating form:
| 1 | Click the ghost form. A new instance of the form opens. |
| 2 | Fill in the form instance and click Save changes. If you have not reached the maximum allowed number of instances of a repeating form, a ghost form will appear every time you save a new instance of the form. If the maximum number of instances of the repeating form has been reached, no ghost form will appear upon saving a new instance of the form. |
To delete an instance of a repeating form:
| 1 | Open the respective instance of the form and click Edit. |
| 2 | Click Delete form. |
| 3 | Select or enter a reason for deleting the form and click Delete. The instance of the form is removed. |
Note!
- It is not possible to reset a repeating form. If you wish to clear the data, it is only possible to completely delete that specific instance of a repeating form and create a new one again.
- In order for the event to be considered as completed, at least one instance of a repeating form has to be filled in.
Repeating item groups
Using repeating item groups allows you to dynamically add multiple rows of data within the same form. This is useful when the number of repeated observations varies, such as multiple measurements or multiple analytes in a single form.
Repeating item groups are defined in the study design. The minimum and maximum number of item groups that can be added is also defined in the study design.
Notes!
- Repeating item groups differ from common event forms, which repeat at the form level. Repeating item groups repeat only a group of items within a single form.
- Repeating item groups can not be used inside a form included in a subject initiated event, or a form set as a randomization or as an allocation form.
- Forms in Viedoc Me do not support repeating item groups.
Adding a repeating item group
To add an instance of a repeating item group to a form:
| 1. |
Open a form that contains a repeating item group: 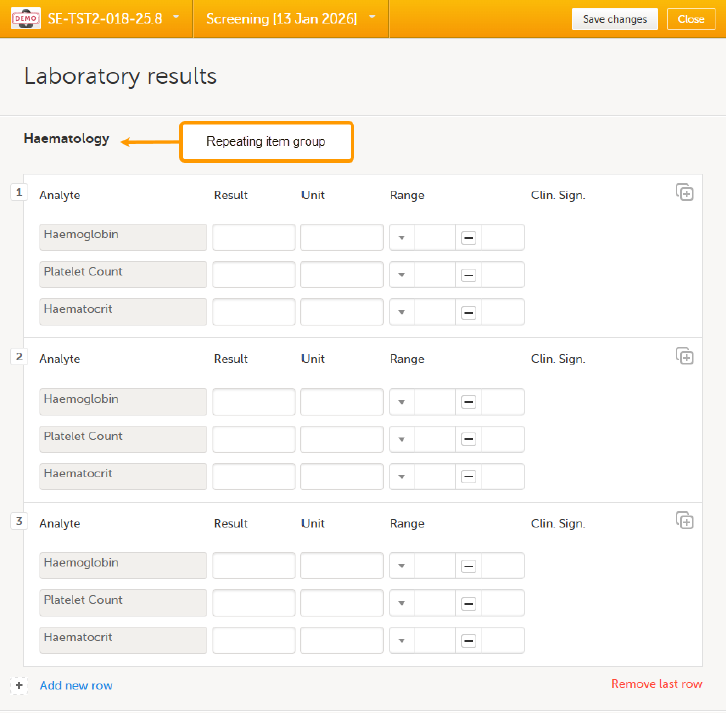
|
| 2, |
Select Add new row:
|
| 3. |
Enter your data and select Save changes. If the maximum number of occurrences has been reached, no additional rows can be added. Notes!
|
Removing the last row of a repeating item group
To remove the last row of a repeating item group:
| 1. |
Open the form and select Remove last row: 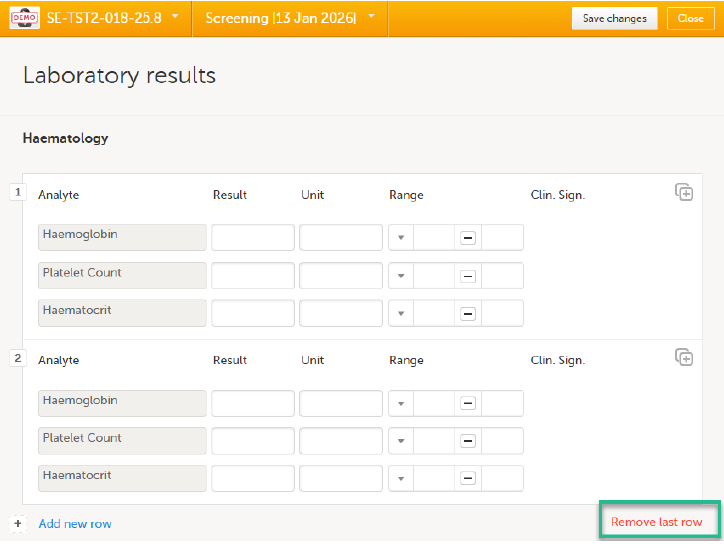
|
| 2. |
Select Save changes. You are prompted for a reason for change: Give reason. 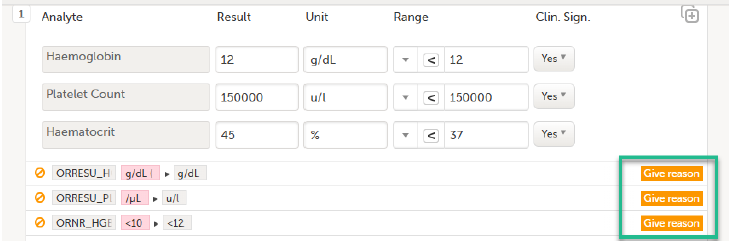
|
| 3. |
The Choose reason for changed value pop-up is displayed: 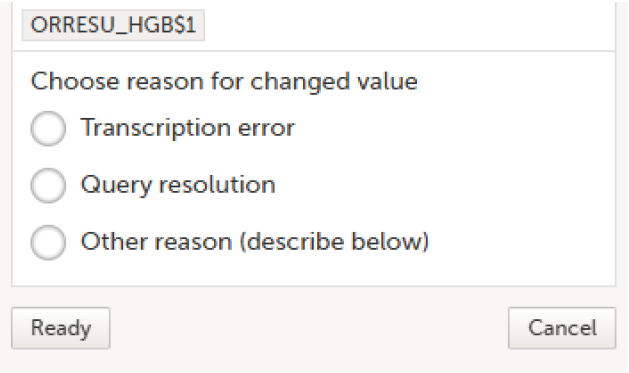
Select an option in the pop-up, for example Query resolution. The same reason is applied to all items in the same instance of that repeating item group: 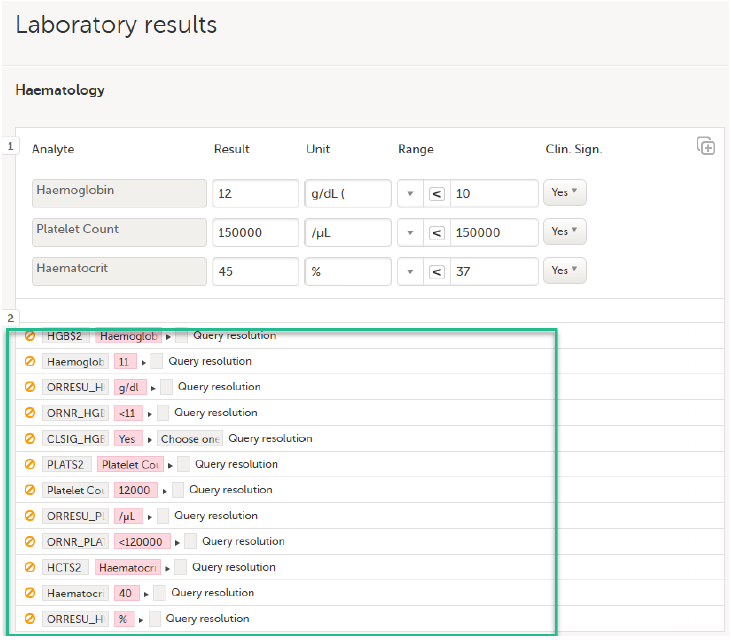
|
Notes!
- If a form is both copyable and contains repeating item groups, all existing instances of the repeating item group from the previous form are copied into the new event when the form is initiated by copying data.
- Adding or removing an instance of a repeating item group is recorded in the audit trail.
- The audit trail is visible in the form history and included in the form history PDF.
Copyable forms
It is possible to initiate a form based on copied data from a previous booklet. This can only be done if the form is set as copyable in the study design.
If a form is set as copyable, a ghost form will appear above the main form.
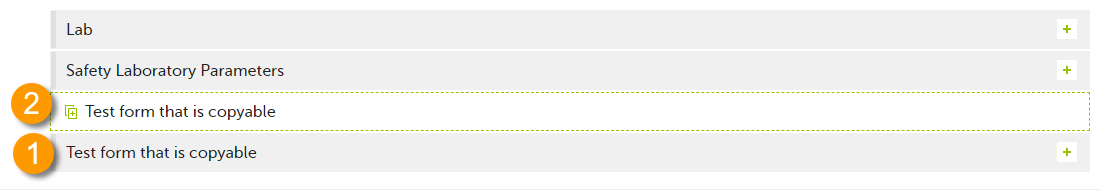
1. Main form - click the main form to enter data manually. Data from the same form in a previous booklet will not be copied into this form in the current booklet.
2. Ghost form - click the ghost form to initiate the form by copying data from a previous booklet. See below for instructions.
Note! The ghost form of a copyable form is displayed above the main form, and marked with a copy icon. If you see a ghost form below the main form, it is a repeating form. See the image below, and see Repeating forms.
1. Main form instance of repeating form.
2. Ghost form of a repeating form, displayed below the main form instance and marked with a + icon.
3. Main form instance of a copyable form.
4. Ghost form of a copyable form, displayed above the main form instance and marked with a copy icon.
If the copyable form is also set as repeating (see Repeating Forms above), and in the previous event, three instances of that form have been initiated, then in the current event all three instances appear as ghost form. By clicking one of these ghost forms, you can select the instance from which the data are copied.
To initiate a form based on data copied from a previous event:
| 1 |
Click the ghost form. 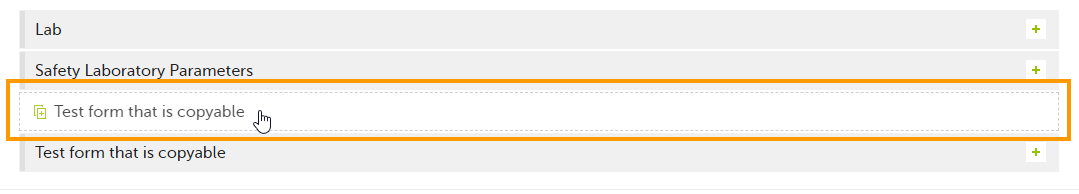
A pop-up opens asking you to confirm whether you want to create a form instance by copying data from a previous event. 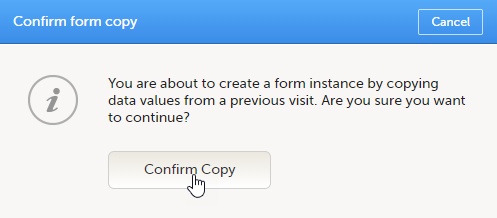
|
| 2 |
Click Confirm to continue. 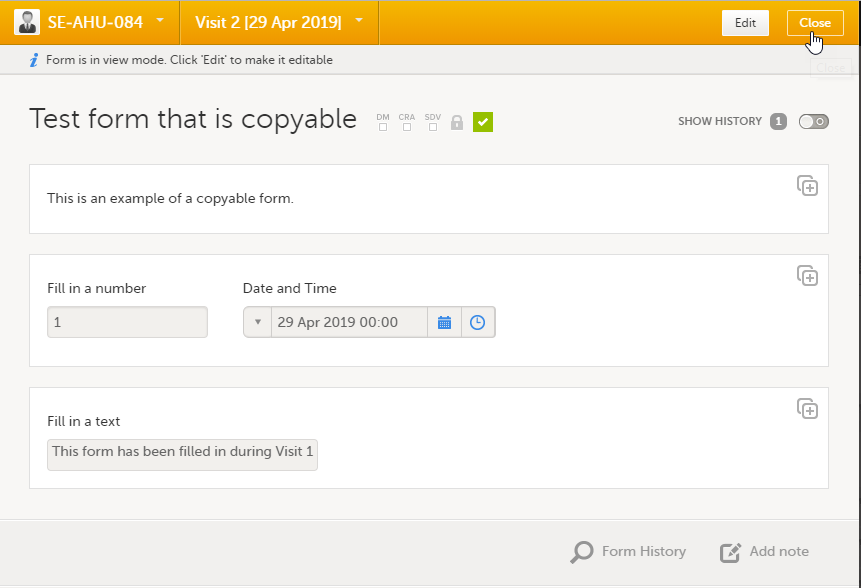
|
| 3 |
The ghost form disappears and the form is displayed as initiated. |
To delete a copied form:
| 1 | Open the form and click Edit. |
| 2 | Click Delete form. |
| 3 | Select or enter a reason for deleting the form and click Delete. The copied form is removed and the ghost form re-appears. |
Note!
- A form instances based on copied data always contains the data of the previous booklet, even if data have been changed during that event. If a copyable form is included in Booklet 1, 2 and 3, and the data are copied from Booklet 1 to Booklet 2, edited during Booklet 2, and then copied to Booklet 3, the changes made during Booklet 2 are included in the data copied into Booklet 3.
- A form instance based on copied data always contains the latest saved data from the previous booklet at the moment of copying. Any changes performed to the form instance from which the data are copied (the source) after the copy action are not reflected in the form instance containing the copied data (the destination).
Confirming data as missing
If you are not able to provide information on a field, you can mark the data as missing, by following the steps below:
| 1 | Click the action icon in the top right corner of each field group: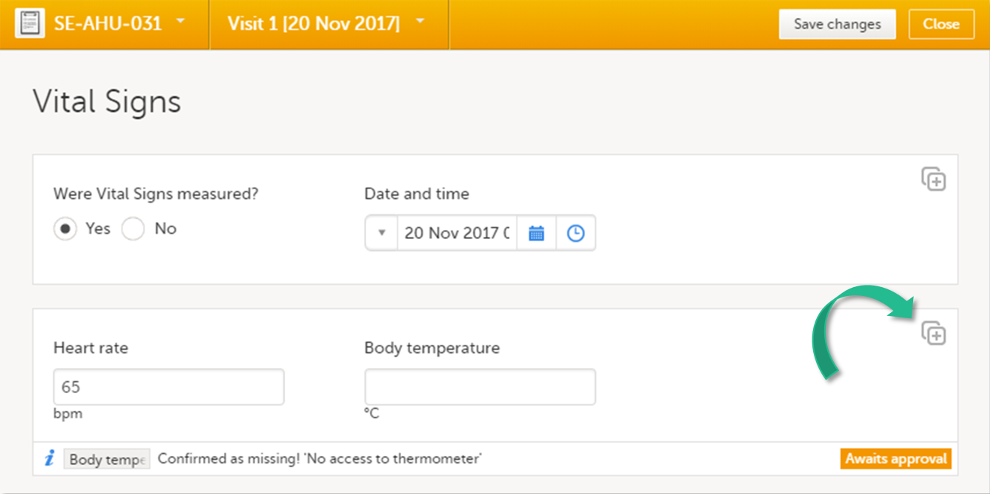 A pop-up opens. |
| 2 | Select the field for which the data is missing in the drop-down menu. |
| 3 | Select Confirm field is missing and provide a reason. |
| 4 | Click Ready. Below the field, the text Confirmed as missing is displayed, together with the reason provided above. |
Private notes
You can add private notes for:
Note! It is possible to add private notes only if this feature is enabled for your role.
Private notes for booklets
You can add private notes to every event.
Private notes are only visible to the user that created the notes, and can only be edited by that user. Adding or editing a private note does not break the signature.
It is possible to add as many notes as you like. Notes are ordered according to date in descending order.
To add a private note for an event:
| 1 | Click Add note on the right pane of the subject details page. |
| 2 | Enter the note text and click Ready. |
| 3 | Click Save changes. The notes pop-up closes. |
To add another note:
| 1 | Click Open notes. |
| 2 | Click Add another note. |
| 3 | Enter the note text and click Ready. |
| 4 | Click Save changes. The notes pop-up is closed and the new note is displayed in the right pane of the subject details page. |
To edit an existing note:
| 1 | Click Open notes. |
| 2 | Click the pen icon behind the note you want to edit. |
| 3 | Edit the note text and click Ready. |
| 4 | Click Close. The notes pop-up is closed. |
To delete a private note:
| 1 | Click Open notes. |
| 2 | Click the trash can icon behind the note you want to delete. |
| 3 | Click Save changes. The notes pop-up closes. |
To see a history of changes to private notes, activate the Show history switch.
Private notes are not recorded in the audit trail, neither included in the data export.
Private notes for forms
You can add private notes to every form.
Private notes are only visible to the user that created the notes, and can only be edited by that user.
Adding or editing a private note does not break the signature.
To add a private note for a form:
| 1 | Click Add note on the bottom of the form. |
| 2 |
Enter the note text and click Save note. The note pop-up closes and the note is displayed on the form. |
Private notes for fields
You can add private notes to single fields in a form.
Private notes are only visible to the user that created the notes, and can only be edited by that user.
Adding or editing a private note does not break the signature.
To add a private note for an event:
| 1 |
Click the action icon in the top right corner of the field group. |
| 2 |
Enter the note text and click Ready. |
| 3 |
Click Save changes. |
Resolving a query
For complete instructions on how to resolve a query, see Resolving queries.
See also:
- The video tutorial Issues: Resolve a query.
- Overview of the queries process and workflow in Viedoc - Queries overview.
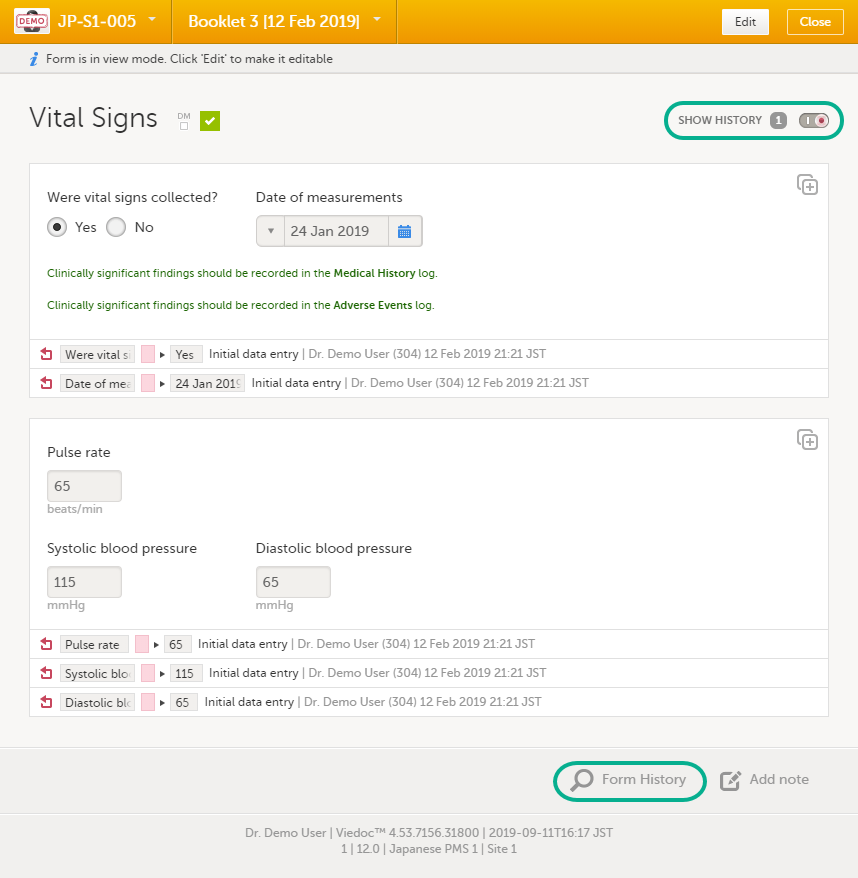
Audit trail and Form History
You can view the history of a form, including information on who entered the initial data and who made any changes. To view the history, activate the Show history switch on the top right side of the form:
Limited number of audit trail records
To make the form history and the form history PDFs more manageable in terms of size, there is a limit to the number of displayed audit trail records. The history only displays the initial data entry and the latest 25 audit trail records. If there are more records, a message is displayed:
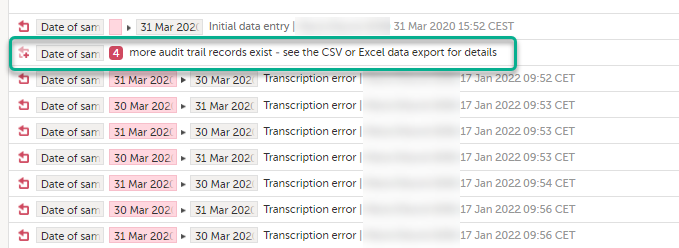
To see the complete form history, export to CSV or Excel.
Note! It is not very likely that clinic staff would make more than 25 edits to a field. However, when working with JavaScript functions, that is a possibility.
Form history PDF
You can download PDFs of all the saved versions of the form by clicking Form History located on the bottom left side of the form.
If any of the fields in the form are hidden for your role, you are not able to see the form PDFs.
By clicking the Form History a list with all the form versions is displayed, and you can choose which version you want to download. There is one version of the form for each change performed on the eCRF.
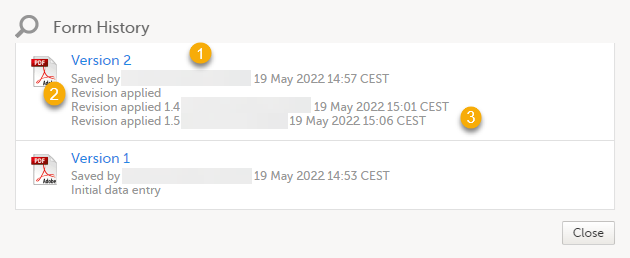
1. The user name of the user who saved the edited form.
2. "Revision applied" is displayed when a user has updated the form according to a new design revision. This can be done in one of two ways:
- Manually edit a specific form
- Manually perform a batch update of all forms with a pending upgrade
3. "Revision applied X.Y <Study Manager user name> <date time>" is displayed when the system has automatically updated the form according to a new design revision. This happens if the update in the revision does not affect the data integrity.
The PDF shows a screenshot of the form with the editing history included:
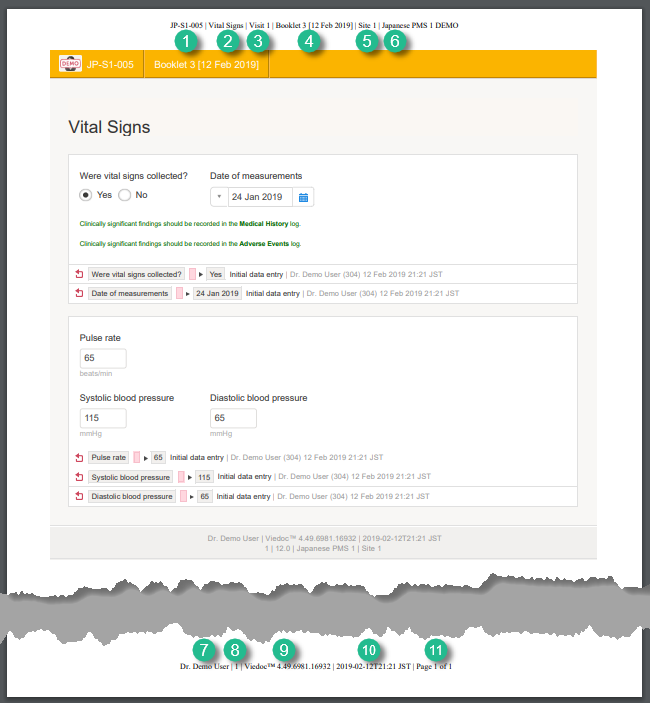
The page header and footer provide the following information, as illustrated in the previous image:
1. Subject ID
2. Form name
3. Event info (in the format set in the study design)
4. Booklet info (in the format set in the study design)
5. Site name
6. Study name
7. User name - the user who has last edited the form
8. Form version number - the version of the eCRF
9. Viedoc version number
10. Date and time when the form was last edited
11. Page number out of total number of pages of the PDF document
Blacklisted file formats
The following executable file types are considered as high risk because essentially all computers with the listed operating system installed can carry out the commands contained in the executable file.
In other words, files with the extensions listed below can be executed with some basic part of Windows, Mac OS, Linux, and so on.
Due to this, the following file formats are not accepted by Viedoc form file loader:
| Extension | Format | Operating system(s) |
|---|---|---|
|
ACTION |
Automator Action |
Mac OS |
|
APK |
Application |
Android |
|
APP |
Executable |
Mac OS |
|
BAT |
Batch File |
Windows |
|
BIN |
Binary Executable |
Windows, Mac OS, Linux |
|
CMD |
Command Script |
Windows |
|
COM |
Command File |
Windows |
|
COMMAND |
Terminal Command |
Mac OS |
|
CPL |
Control Panel Extension |
Windows |
|
CSH |
C Shell Script |
Mac OS, Linux |
|
EXE |
Executable |
Windows |
|
GADGET |
Windows Gadget |
Windows |
|
INF1 |
Setup Information File |
Windows |
|
INS |
Internet Communication Settings |
Windows |
|
INX |
InstallShield Compiled Script |
Windows |
|
IPA |
Application |
iOS |
|
ISU |
InstallShield Uninstaller Script |
Windows |
|
JOB |
Windows Task Scheduler Job File |
Windows |
|
JSE |
JScript Encoded File |
Windows |
|
KSH |
Unix Korn Shell Script |
Linux |
|
LNK |
File Shortcut |
Windows |
|
MSC |
Microsoft Common Console Document |
Windows |
|
MSI |
Windows Installer Package |
Windows |
|
MSP |
Windows Installer Patch |
Windows |
|
MST |
Windows Installer Setup Transform File |
Windows |
|
OSX |
Executable |
Mac OS |
|
OUT |
Executable |
Linux |
|
PAF |
Portable Application Installer File |
Windows |
|
PIF |
Program Information File |
Windows |
|
PRG |
Executable |
GEM |
|
PS1 |
Windows PowerShell Cmdlet |
Windows |
|
REG |
Registry Data File |
Windows |
|
RGS |
Registry Script |
Windows |
|
RUN |
Executable |
Linux |
|
SCR |
Screensaver Executable |
Windows |
|
SCT |
Windows Scriptlet |
Windows |
|
SHB |
Windows Document Shortcut |
Windows |
|
SHS |
Shell Scrap Object |
Windows |
|
U3P |
U3 Smart Application |
Windows |
|
VB |
VBScript File |
Windows |
|
VBE |
VBScript Encoded Script |
Windows |
|
VBS |
VBScript File |
Windows |
|
VBSCRIPT |
Visual Basic Script |
Windows |
|
WORKFLOW |
Automator Workflow |
Mac OS |
|
WS |
Windows Script |
Windows |
|
WSF |
Windows Script |
Windows |
|
WSH |
Windows Script Preference |
Windows |