Deleting a study (for Study Manager)
This lesson describes how a study is deleted. The instructions are intended for the Study Manager (STM).
Introduction
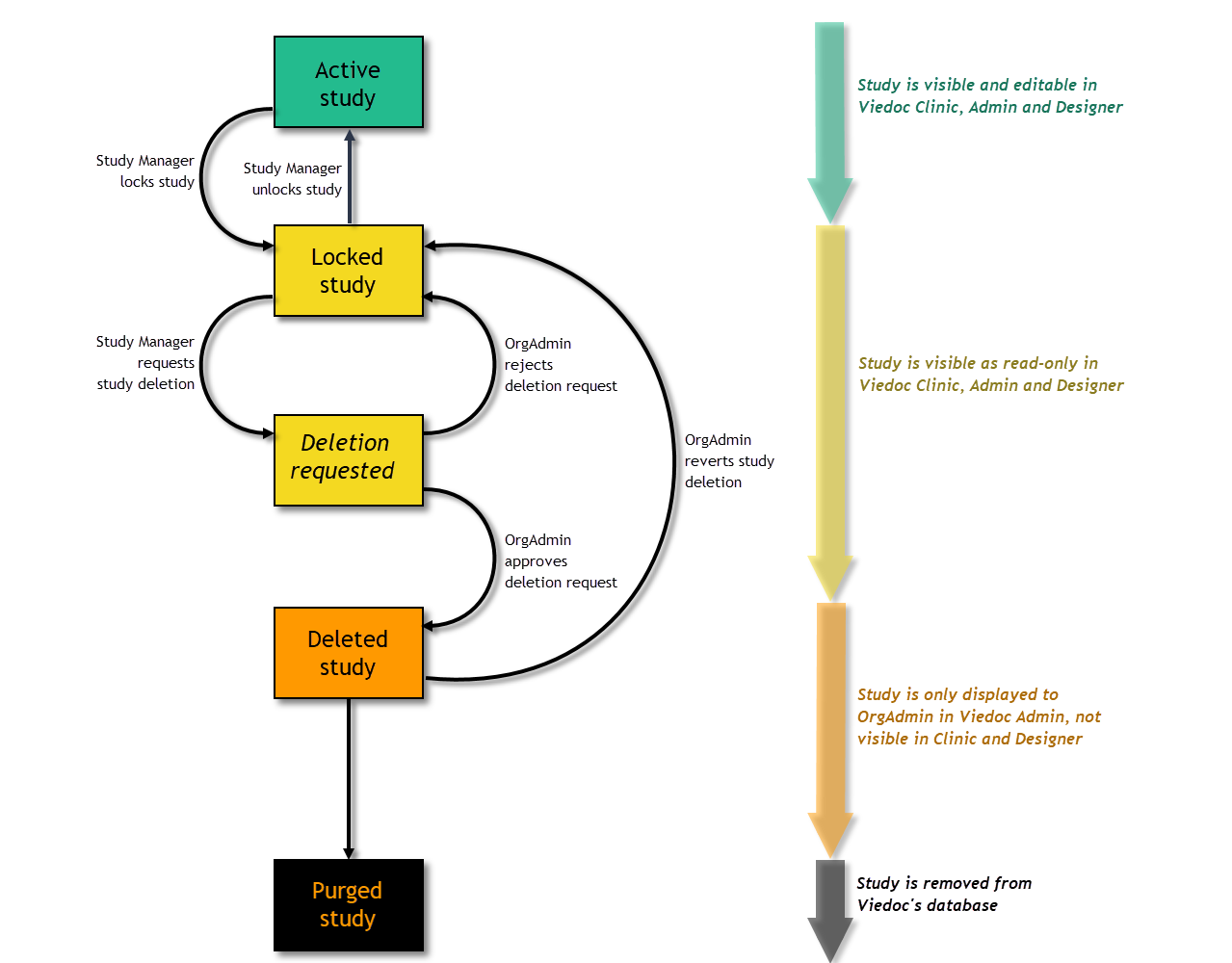
A study can be permanently deleted from Viedoc when the study is locked. Deletion is initiated by the Study Manager, who can submit a request to delete the study from Viedoc to the Organization Administrator. The Organization Administrator can then approve or reject a request for study deletion.
After study deletion is requested by the Study Manager and approved by the Organization Administrator, the study is shelved on Viedoc's database. A deleted study is not visible in Viedoc Clinic or Viedoc Designer, the study is only displayed in the study overview page of the Organization Administrator in Viedoc Admin.
The Organization Administrator is able to revert the deletion of a study within 180 days after the deletion request has been approved. After this period, the study will be permanently purged from Viedoc's database, and all study details and data will be permanently removed. It will not be possible to find any traces of the study and the subjects included.
For traceability, all study delete actions are audit trailed. You can download a report that provides a full history of all requests for study deletion, approvals of study deletion, and reversions of study deletion that are performed in the study, including who performed the actions and when (date and time in UTC), and the reason that was given for deleting the study or reverting study delete. The report also contains the full history of all lock and unlock actions.
Note! This section is intended for the Study Manager. For instructions for the Organization Manager, see Deleting a study (Org Admin).
Step-by-step guides - for the Study Manager
Requesting study deletion
A request for study deletion can only be submitted by the Study Manager. A study can only be deleted after the study is locked. For information on how to lock a study, see Locking a study.
Note! Before deleting the study, make sure that you have downloaded the user report, the data export archive and the study design. Download and archive the study data in the formats necessary, available formats are Excel, Comma-Separated Values (CSV), Operational Data Model (ODM) and PDF. For instructions, see:
- Downloading the user report in Managing Users (STM and SIM) (Viedoc Admin),
- Exporting data (Viedoc Clinic),
- Exporting/Locking/Deleting a study design (Viedoc Designer).
To submit a request for study deletion:
| 1 | Open the study in Viedoc Admin and select Study settings. The Study settings pop-up opens. |
| 2 |
Select the blue pen icon. The study status pop-up opens. |
| 3 | Select Request study deletion. The request study delete pop-up opens. |
| 4 |
Select whether the following actions are done or not done:
Enter a reason for deleting the study, and enter your password. 
|
| 5 | Select Request study deletion. The Study status page displays that deletion of the study is requested, by whom and when (date and time in Coordinated Universal Time (UTC)), and the study delete request will be sent to the Organization Administrator. All Study Managers and Organization Administrators will be notified of the request by email.  |
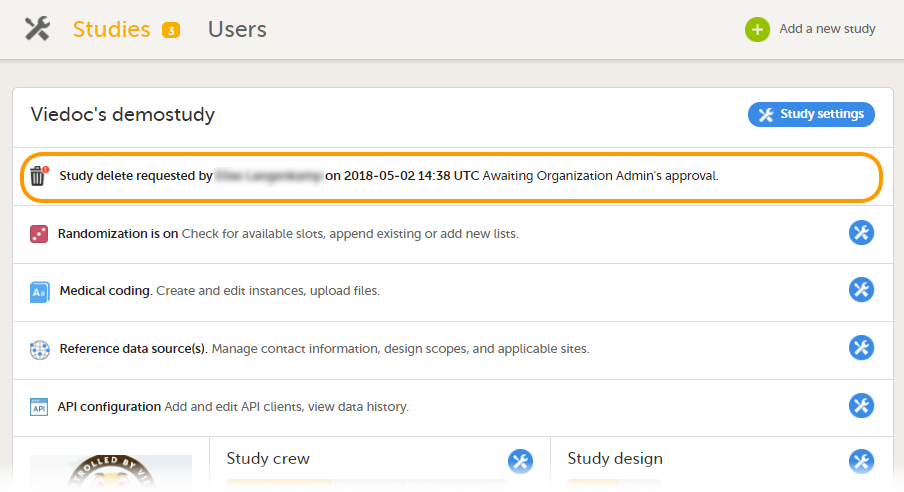
When study deletion has been requested, the study page will display the status "Study delete requested by ... on ..." Until the request has been approved, the study will be in locked state and visible in Viedoc Clinic and Viedoc Designer.
Downloading the study status report
To download the study status report:
| 1 | Open the study in Viedoc Admin and select Study settings. The Study settings pop-up opens. |
| 2 |
Select the blue pen icon. The Study status pop-up opens. |
| 3 | Select Download study status report. A PDF is downloaded that lists all database lock and delete actions, including when and by whom the study was locked/deleted, and the reason that was given for locking/deleting the study. |
